Introduction
Immune response to implants is commonly reported in the literature and can include hypersensitivity related to pacemakers, dental implants, and orthopedic hardware.1 Furthermore, up to 13% of people are sensitive to nickel, cobalt, or chromium.1, 2, 3, 4, 5, 6, 7, 8, 9 The development of metal sensitivity after implantation of orthopedic hardware is common.1, 10, 11, 12
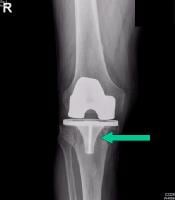
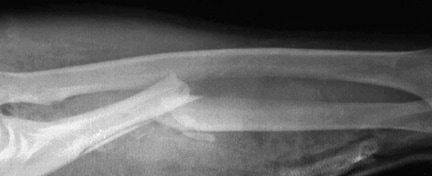
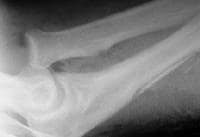
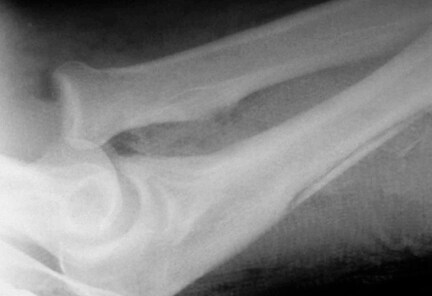
Osteolysis around a total knee implant.
Osteolysis around a total knee implant.
Metal sensitivity is also correlated with osteolysis and aseptic loosening of implanted metal hardware.
5, 10, 12, 13, 14, 15, 16, 17, 18 However, statistical reviews of cases involving adverse reactions after implantation of metal hardware demonstrate that metal sensitivity can be proven causative in less than 0.1% of cases in which sensitivity reactions exist.
1, 19, 20, 21, 22 Therefore, the clinical significance of metal sensitization remains a question. Nonetheless, the orthopedic surgeon must be aware of the potential problem, and when other more common causes of implant failure have been excluded, the possibility of allergic reaction to the metal must be considered, evaluated, and treated.
According to Huber et al, the presence of corrosion products and a hypersensitivity reaction in patients suggests that there is a relationship between corrosion and implant-related hypersensitivity. In 11 cases in which periprosthetic tissue contained corrosive elements (solid chromium orthophosphate corrosion products) after aseptic loosening of articular implants, immune-response tissue reactions were identified in all 11 cases.
23For excellent patient education resources, visit eMedicine's
Foot, Ankle, Knee, and Hip Center. Also, see eMedicine's patient education articles
Total Hip Replacement and
Knee Joint Replacement.
Related eMedicine topics:
Implantable Cardioverter-Defibrillators
Total Knee Arthroplasty
Arthroplasty Component Failure
Arthroplasty-Associated Infections
Immunosuppression
Literature Review
This issue of the clinical significance of sensitization to implanted metals has long been a matter of debate in the literature. While some studies have shown that sensitization to metal implants is prevalent,1, 10, 11, 12 others refute these data and have concluded that not only does hypersensitivity fail to develop24; but patients with metal hypersensitivity prior to implantation actually become desensitized and anergic after implantation.25 Some authors have further suggested that metal hypersensitivity may be associated with bone loss and aseptic loosening of implanted devices,5, 11, 14 while other authors have concluded that even if a metal allergy exists, no adverse effects occur.13, 24, 26, 27
These contradicting studies make it difficult for the orthopedic surgeon to make the diagnosis of symptomatic metal allergy with confidence. The confusion could be the result of the presence of different metals in the implants, different manufacturing methods, small numbers of patients in the studies, and difficulty in achieving adequate diagnosis. More research is clearly needed. In the meantime, the diagnosis must be made with caution. Infection, nonunion, aseptic loosening, other inflammatory conditions, mechanical failure of the implant, and malalignment issues must be excluded first before assuming the problem is an allergic reaction.
Workup And Diagnosis
Clinical presentation
The presenting signs and symptoms of a nickel hypersensitivity to an implanted orthopedic device are variable but usually consist of the expected complaints of a patient with hardware failure. Patients with joint replacements have symptoms of loosening, including pain and instability. (For example, with a total hip replacement, the patient often has groin pain radiating to the medial thigh.) Patients with hardware for fractures have symptoms of nonunion, including pain and motion at the fracture site. Local inflammatory symptoms similar to the symptoms of infection are also possible, including warmth, erythema, and swelling over the implant, although systemic complaints such as fever are unlikely. A skin rash may develop over the metal device but is not always present.28, 29, 30
Osteolysis due to implant loosening is always in the differential diagnosis. The mechanism of osteolysis is primarily a local reaction to particulate debris,31 which leads to a cascade of cellular reactions (including activation of monocytes/macrophages, phagocytosis, release of cytokines) that eventually lead to increased osteoclastic activity around the prosthesis. Osteolysis is a reaction to local irritation, not an immune hypersensitivity response.32
Workup
Blood tests
Currently, blood tests are rarely performed in the workup of immune responses to implants. Generally, white blood cell counts and other inflammatory mediators (such as platelet counts, C-reactive protein levels, and sedimentation rates) are not elevated, or are only minimally elevated, and they are not specific or reliable enough to help in the diagnosis.
Radiologic studies
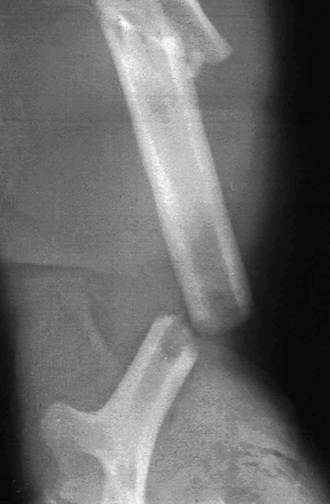
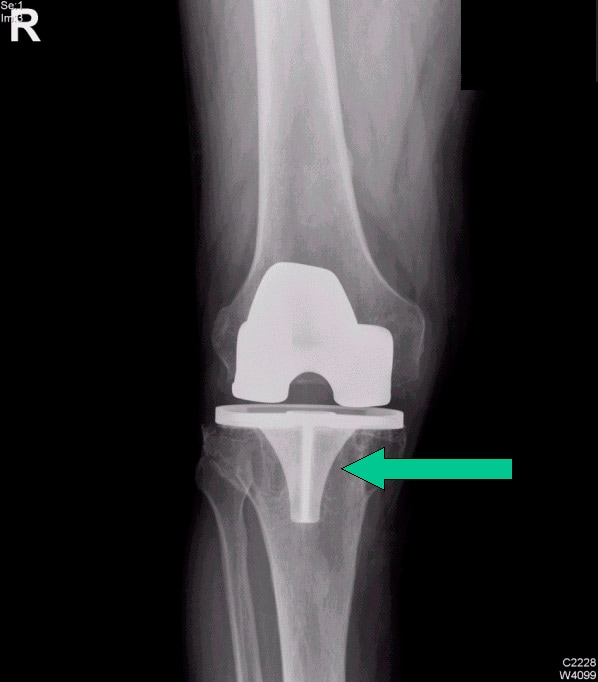
Careful radiographic assessment of the implant is required. Radiolucencies around the hardware, screw migration, and change in position of the implant imply loosening that could be due to hypersensitivity to the metal. Cystic changes, as seen in osteolysis, may be seen (see Image below and Image 1 in Multimedia). CT scans and MRIs are not especially helpful. A bone scan may be positive, but the findings are nonspecific.

Osteolysis around a total knee implant.

Osteolysis around a total knee implant.
Skin patch testing
Traditionally, skin patch testing has been the standard screening test for metal hypersensitivity. This method is limited by the fact that a positive result is not indicative of a true hypersensitivity but must be considered in the context of a patient's medical history and physical findings.33, 34 A routine skin test reveals a prevalence of metal sensitivity of 0.2% for chromium, 1.3% for nickel, and 1.8% for cobalt. After placement of metal implants, sensitization (change from negative to positive) occurs in 2.7% for chromium, 3.8% for nickel, and 3.8% for cobalt. Desensitization (change from positive to negative) occurs in 0% for chromium, 2.1% for nickel, and 3.8% for cobalt.35
Many patients with implanted metal hardware have positive skin test results for those metals but are completely asymptomatic.13, 24 If the skin patch test finding is positive, the patient can be designated allergic. However, the clinical significance of the allergy is controversial. Most allergy skin patch tests that show skin reactivity have no clinical implications.
The causes of these skin immunologic reactions are unclear.1, 34 It is thought that antigen-presenting cells that are localized to the skin (dendrite cells) may handle antigens differently than the antigen-presenting cells that are systemic (macrophages and monocytes). Therefore, many people who have skin reactivity to metals may never develop any reactivity at the site of prosthesis composed of that metal. Skin test results may not return to normal after metal removal.16 Conclusions based on skin patch testing should therefore be made with caution and only assumed valid if the whole clinical picture supports the finding of symptoms related to metal allergy. Preoperative skin patch testing is not typically recommended unless there is a strong suggestion of established sensitivity by history, because of a slight chance of sensitization and the high cost/low yield results expected.
Lymphocyte transformation test
Tests that may be more specific include the lymphocyte transformation test (LTT) and the lymphokine migration inhibition factor (MIF) test, which have been used to help diagnose metal hypersensitivity.
In vitro LTT proliferation testing is perhaps the most prevalent method after patch testing for assessing hypersensitivities. It involves measuring the proliferative response of lymphocytes following activation. A radioactive marker is added to lymphocytes along with the desired challenge agent. The incorporation of radioactive H3 -thymidine marker into cellular DNA on division facilitates the quantification of a proliferation response through the measurement of amassed radioactivity after a time period of 3-6 days. At day 6, H3 -thymidine uptake is measured by using liquid scintillation. The proliferation factor or stimulation index is calculated by using measured radiation counts per minute (cpm), as follows:
Proliferation factor = (mean cpm with treatment)/(mean cpm without treatment).
The use of proliferation testing in the assessment of metal sensitivity has been well established as a method for testing metal sensitivity in a variety of clinical settings.36, 37, 38, 39, 40, 41 The technical sophistication and high expense of LTT testing for implant-related metal sensitivity has limited its use; therefore, few conclusions can be drawn.42, 43, 44 These few investigations using LTT testing report that increased rates of metal sensitivity can be detected above that determined by dermal patch testing.42, 44, 45 Such reports seem to indicate LTT testing may be equally or better suited for the testing of implant-related sensitivity than dermal patch testing.37, 38, 39, 40, 41, 42, 43, 44
Lymphokine migration inhibition factor test
Another in vitro test that has shown promise in diagnosing metal hypersensitivity involved the use of MIF. MIF acts to prevent lymphocytes from leaving a site where foreign antigens are present. This test selectively detects lymphokine MIF, which, when present, does indicate an active immune response and metal sensitivity.26
The test is performed by obtaining a blood sample and isolating the lymphocytes. The lymphocytes are then mixed with solutions of specific metal ions, such as nickel, chromium, cobalt, or titanium. The test result is considered positive if the lymphocytes stay in the metal ion solution, indicating a cellular reaction to the metal dissolved in that solution. (In a positive result, no migration occurs.) The test result is considered negative if the lymphocytes migrate away from the particular metal ion solution, indicating that they are not reacting to the dissolved metal.1
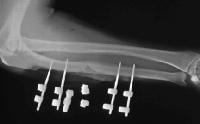
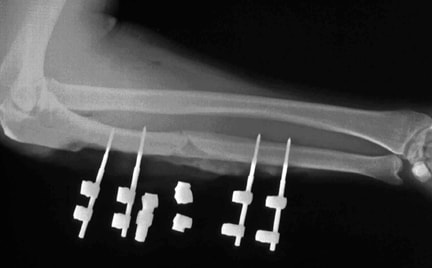
Studies reveal that positive MIF test results to metals implanted in an orthopedic patient are well correlated with pain, swelling, and dermatological reactions over that area. Furthermore, after the implanted materials are removed, these signs and symptoms improve and the MIF test result returns to normal.16 The lymphokine MIF is the most useful clinical test for diagnosis of hypersensitivity reaction to orthopedic implants (see Image below and Image 2 in Multimedia).46 Cement wear particles are immunologically inert and have specifically been found not to cause a lymphocyte response in vitro,47 so the lymphokine MIF result should be negative in osteolysis.
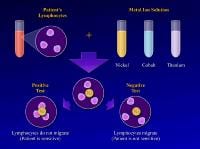
(Click Image to enlarge.) Lymphokine migration test.
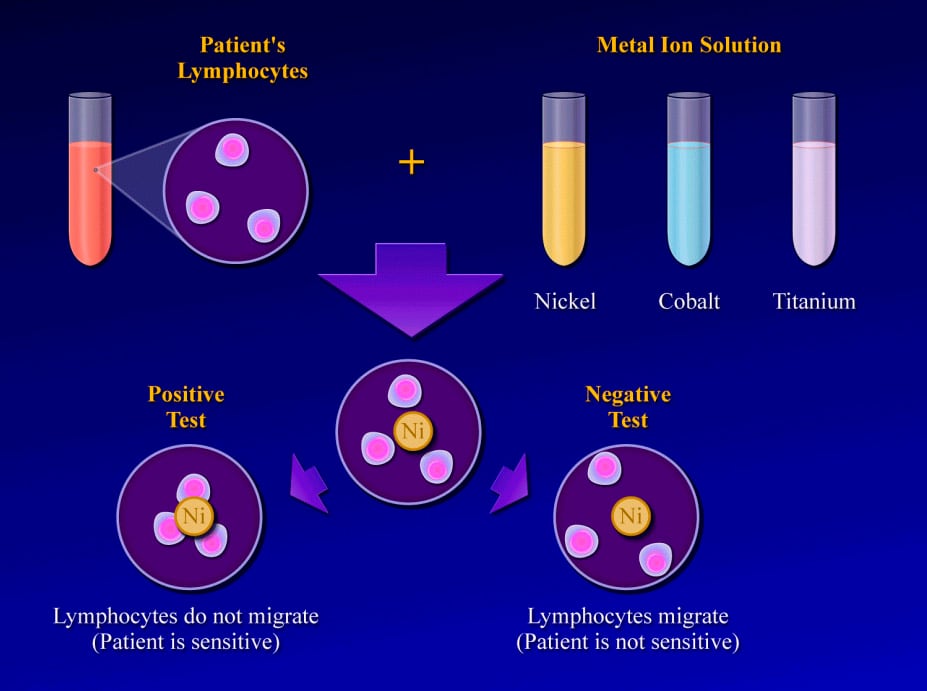
(Click Image to enlarge.) Lymphokine migration test.
Case Example
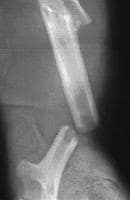
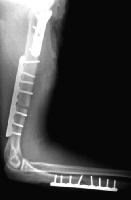
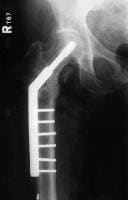
A 71-year-old woman had a right intertrochanteric hip fracture and underwent open reduction and internal fixation (ORIF) by using a standard stainless steel hip fracture implant (Synthes DHS; Paoli, PA). Postoperatively, the patient did well, with evidence of fracture healing, full weight bearing, and full range of motion by 3 months after surgery.
Approximately 6 months later, the patient began to complain of right hip pain laterally over the area of the implanted hardware. The fracture was radiologically healed, but, because of the patient's unbearable pain, a technetium bone scan and tomogram of the area were obtained. The results demonstrated increased uptake and lucency around the lag screw, indicative of hardware loosening.
The patient underwent debridement, hardware exchange, and an iliac crest bone graft. Intraoperative cultures were obtained that all proved negative for an infectious cause. Again, 4 months postoperatively, the patient began to complain of similar right hip pain, although images showed good bone graft incorporation and fracture healing (see Image below and Image 4 in Multimedia).